Welcome to Aptium’s (slightly delayed) quarterly update. The last 4-5 months have been incredibly busy, with a number of significant milestones achieved which this update will highlight. The most exciting milestone however, has been validating the significant demand for the Company’s flagship scanner, MultiScan4D™. We were able achieve this important milestone after receiving our first 5 evaluation units from our manufacturing partner IntelliDesign.
Since the last update Aptium has attended and displayed at multiple international trade shows. The Company has also engaged directly with end-users who have been evaluating the MultiScan4D™ in their facilities. The feedback has been universally excellent to the extent that we have established immediate demand for 130 units with a value of approximately AUD$1 million.
This demand has significantly boosted Aptium’s capital raising efforts leading to advanced negotiations with a number of Family Offices, High Net Worth Individuals, Angel Investors and Venture Capital Funds. To recap our capital raise Aptium is seeking AUD$5 million in equity investment. These funds will enable the Company to:
Finalise our production pipeline with our manufacturing partner IntelliDesign.
Build inventory to fulfil the existing and future demand.
Finalise the development of the MotionScan4D™ and ThermalScan4D™.
Fund ongoing and future sales and marketing activities.
MultiScan4D™
Aptium's Executive Chairman, Mel Bridges, receiving the company's first production MultiScan4D™
Aptium Quarterly Update April 2026


Promotional Activities
On the promotional front Aptium has presented at a number of local and international events. In Australia the company attended and displayed at:
Pedorthic Association Meeting - October 2025, in addition to displaying at this meeting Alex Terrill, our resident podiatrist, presented an oral publication titled “Evaluation of personalised 3D printed functionally graded metamaterial foot orthoses with an offloading boot”
Diabetes Feet Association (DFA) Conference – November 2025 in addition to displaying at this meeting Dr Laith Alzubaidi delivered an oral presentation on "Better Feet, Better Lives: A Portable AI Platform for Early Detection, Monitoring, and Prevention of Diabetic Foot Complications".

Aptium's resident podiatrist Alex Terrill demonstrating the MultiScan4D™ at the Pedorthic Association Conference.
Internationally the company attended and displayed at:
Medica Germany – one the world’s largest trade fairs for Medical Technology and Healthcare – November 2025. While displaying at this event, Aptium was able to showcase our technology to a significant global audience. We were able to validate interest at an end-user level, but more importantly, we were able to establish numerous potential distribution partners in Europe and Latin America.
World Healthcare Exhibition 2026 (WHX) in Dubai, UAE – world’s largest healthcare event – February 2026. Displaying at this event enabled Aptium to further leverage global distribution opportunities.
BioAsia 2026 in Hyderabad India - in conjunction he with the Queensland trade delegation to India, Aptium displayed at this event. India is becoming a major focus for Aptium, in particular for the ThermalScan4D™, as diabetes and the consequential diabetes related foot ulcers has reached epidemic proportions in India. Access to the ThermalScan4D™ provides the Indian healthcare system with a realistic solution to the early identification and intervention to this major health problem.


Aptium's team at WHX, Dubai.
Aptium's team at BioAsia, Hyderabad, with Mary Cunningham from Austrade.
Expanding the Team
To support Aptium’s rapid growth we continue to build out our team. Since our last update we have welcomed three new team members: Ollie Nagle, Lead Engineer & Software Specialist; Denisse Dizon, Quality Software Engineer and Jonn Dillon, Lead Hardware Engineer. These three individuals are already making a significant impact on Aptium’s progress.
Building a Quality System
Building an appropriate Quality Management System is a critical piece of infrastructure that will enable Aptium to leverage the current and ongoing success of our technology. To facilitate this strategic requirement, we have implemented an ISO13485 compliant Quality Management System (QMS). This system will streamline regulatory compliance, enhance product safety and reduce operational costs through automated workflows. The system enables superior real-time control over documentation, accelerates audit readiness, improves data driven decision-making, and facilitates faster global market access. All of Aptium’s technology is designed and developed under ISO 13485, ISO 14971 and IEC 62304, creating a comprehensive, risk-driven, and traceable quality framework. This framework improves device quality by ensuring safety, consistency and compliance from conception to post market surveillance. While ISO 13485 provides the overall Quality Management System (QMS), ISO 14971 handles the risk management methodology, and IEC 62304 provides the specific, rigorous processes required for software development.
Setting Milestones - Measuring our Future
During the last quarter Aptium has built revenue model that will help highlight why we believe the Company represents a great investment in the future. The chart below highlights what Aptium believes will be the major inflection points in the Company’s growth for the next 5 years.
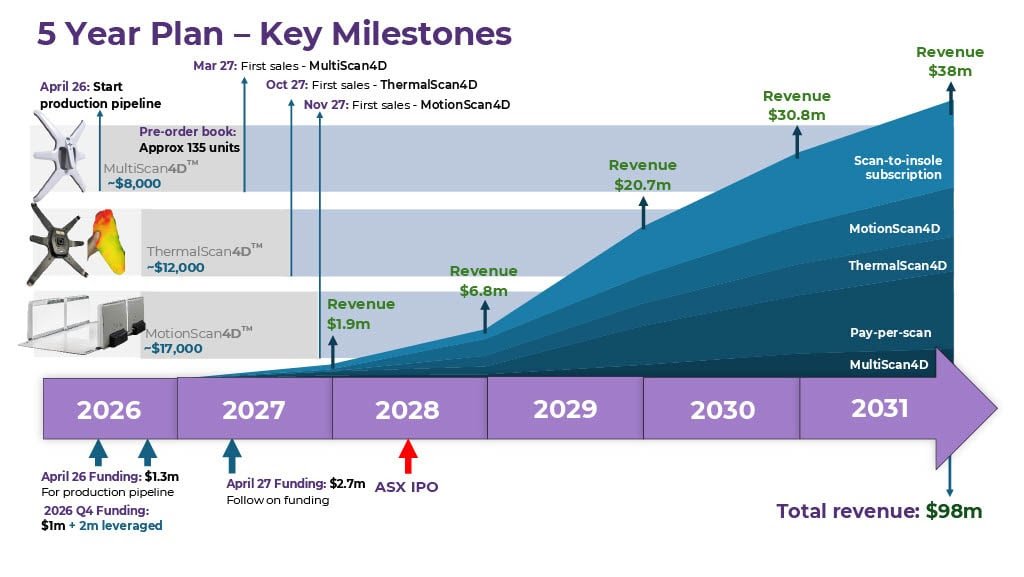

Aptium is well positioned to drive significant revenues during the next 1-2 years with an expectation of becoming cash flow positive in the in the first half of 2028. The Aptium team believe value inflection points outlined above demonstrate a significant investment opportunity.
For investment enquiries, please contact:
Carl Stubbings – carl.stubbings@aptium.ai
Mel Bridges – mel.bridges@aptium.ai
